Research: Eiger Biopharmaceuticals
I’ve owned Eiger for a while. I bought more when they announced their first (disappointing I guess) COVID results in mid-September and the stock tanked. It was silly because this isn’t a COVID stock. But then the second set of results came out today from a COVID study in Toronto and they look very good (see end of post tweets from biotech twitter). I don’t know why the stock wasn’t up more today, given where others go when good COVID results come out. I bought even more today when it was in mid-$9s. Its still below where it was a couple months ago and again, COVID isn’t the main feature here. Notes below and COVID notes at the end.
- Market cap of $300mm
- $60mm of cash
- here is what they are targeting:
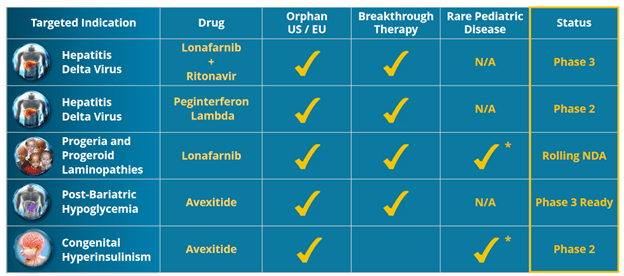
- going after rate and ultra-rare diseases
- 3 drug candidates: Lonafarnib, interferon Lambda, Avexitide – but really the story is about Lonafarnib and Lambda and the Phase 3 studies respectively
- They’ve kinda repositioned their investor presentation from being “rare diseases” to being HDV leader – so its all about Lonafarnib and Lambda
- they are a leader in HDV – Hepatitis Delta Virus
- most severe form of hepatitis
- 4-6% of HBV patients co-invected with HDV
- No FDA approved Rx
- 15mm to 20mm patients worldwide – orphan market in US is >100,000, in West EU is >200,000
- HDV advances much faster than other forms
- HDV is very expensive to care for:

- targeting with interferon-lambda to reduce HDV-RNA and Lonafarnib
Lambda
- LIMT was their Phase 2 Interferon-lambda study:

- results were good for the 180mg dose:
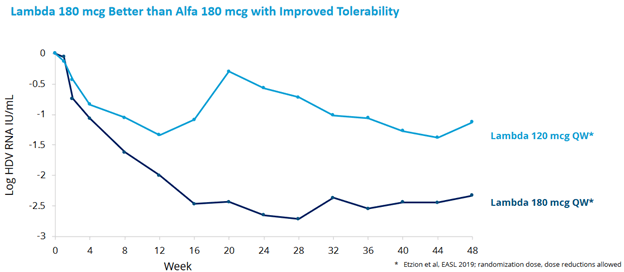
- Lambda on track for approval in next 2-3 years
- Phase 3 study will have 48 week treatment, 24 week post-treatment
- single 180mg dose, looks like design of trial uses Lambda in both wings, just delays by 13 weeks in the one, then compares the 13 week non-use cohort with the end point of the Lambda patients – kinda odd
Ionafarnib
- completed NDA submission for lonafarnib for the treatment of Progeria and Progeroid Laminopathies.
- FDA previously granted Breakthrough Therapy Designation and Rare Pediatric Disease Designation to lonafarnib, which enables eligibility for Priority Review, if relevant criteria are met. Eiger expects to hear from the agency regarding submission acceptance and Priority Review within 60 days.
- Lonafarnib licensed from Merck
- with Lonafarnib – dosing was figured out in Phase 2
- response in Phase 2:
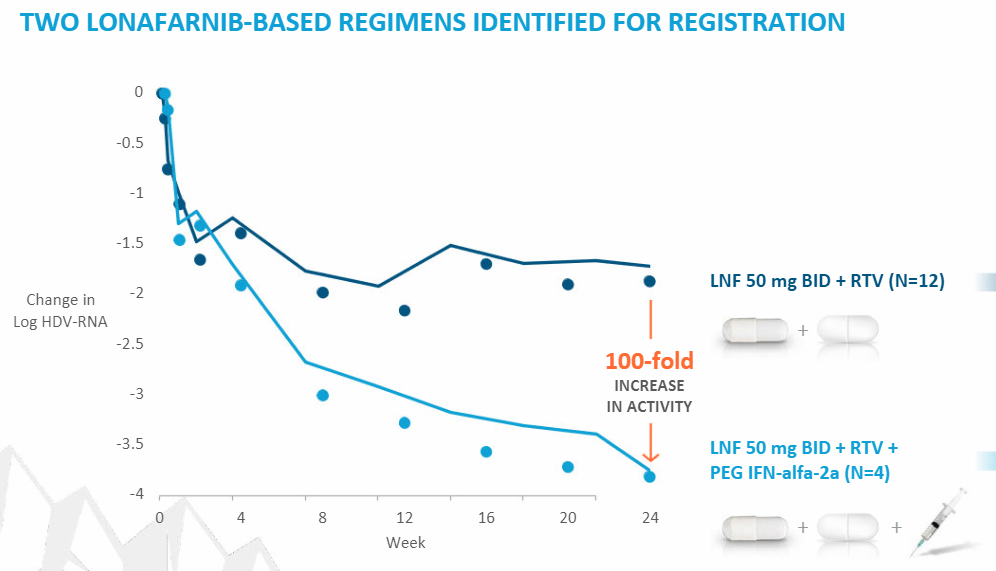
- but what they seem to have found with Lonafarnib is it works best in combo with Ritonavir and IFN-alfa
- Now onto phase 3 study
- the phase 3 has 3 wings, one with the 3-combo, one with 2-combo, one with just IFN-alfa
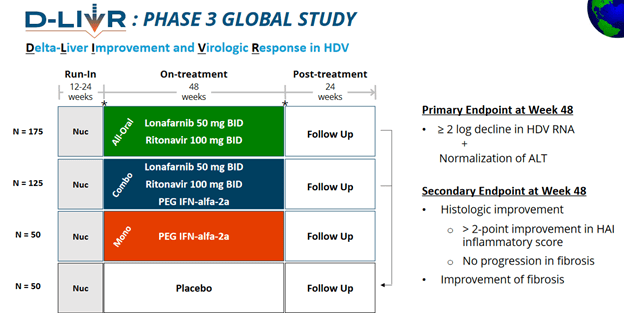
- expect to complete enrollment in 2021
- 48 week treatment, 24 week post-treatment
Lambda for COVID
- multiple studies in parallel
- looks like 6 studies in total
- Toronto, Stanford, Israel, Boston, NY, Baltimore
First Study
- this study looked at use in mild covid – “in outpatients with mild and uncomplicated COVID-19”
- A total of 120 patients were randomized 1:1 to a single subcutaneous dose of Lambda or normal saline placebo. Patients were followed for 28 days.
- this study was reported Sept 28:
The primary endpoint was duration of viral shedding, determined by time to first of two consecutive negative tests for SARS-CoV-2 by qRT-PCR. The secondary endpoint was reducing duration of symptoms and hospitalization in patients with mild COVID-19.
No difference was demonstrated in duration of SARS-CoV-2 viral shedding and time to symptom resolution when compared with placebo. Median time to cessation of viral shedding in both groups was 7 days. Lambda was well-tolerated with few adverse events, which included elevated transaminases which self-resolved.
- this isn’t really surprising, as they said “”We now know that untreated patients with mild COVID-19 clear virus quickly. Published reports have demonstrated evidence of a therapeutic benefit of interferons in hospitalized patients with more advanced COVID-19 disease,”
Second study reported Oct 15
- mild to moderate covid
- said “total of 60 patients were randomized 1:1 to a single subcutaneous dose of Lambda 180 mcg or normal saline placebo. Patients were followed for 14 days.”
- these are the results, seem significant:
SARS-CoV-2 RNA viral load decline from baseline was significantly greater in the Lambda group than in the placebo group from Day 5 onwards. After controlling for baseline viral load, those treated with Lambda were 4.1-fold (95% CI 1.2-16.7, p=0.029) more likely to clear by Day 7 than those in the placebo arm. For those with baseline viral load > 6 log copies/mL, the proportion negative at Day 7 in the Lambda group was 15 of 19 (79%) compared to 6 of 16 (38%) in the placebo group (p=0.013). This difference translated into a median time to clearance of 7 days with Lambda compared to 10 days in the placebo group (p=0.038).
- also this: Lambda works particularly well in patients with high baseline viral loads
Tweets: